Understanding ESR1 Mutations in Advanced Breast Cancer

Although ER blockage confers durable responses, treatment resistance and failure does occur in around 40% of patients with metastatic breast cancer (mBC), resulting in disease recurrence and progression.5 The most common form of treatment resistance stems from the acquisition of point mutations in the ligand-binding domain (LBD) of the ESR1 gene,6 resulting in constitutive activation of the ER even in the absence of ER-bound estrogen.4
The Emergence of the Mutation
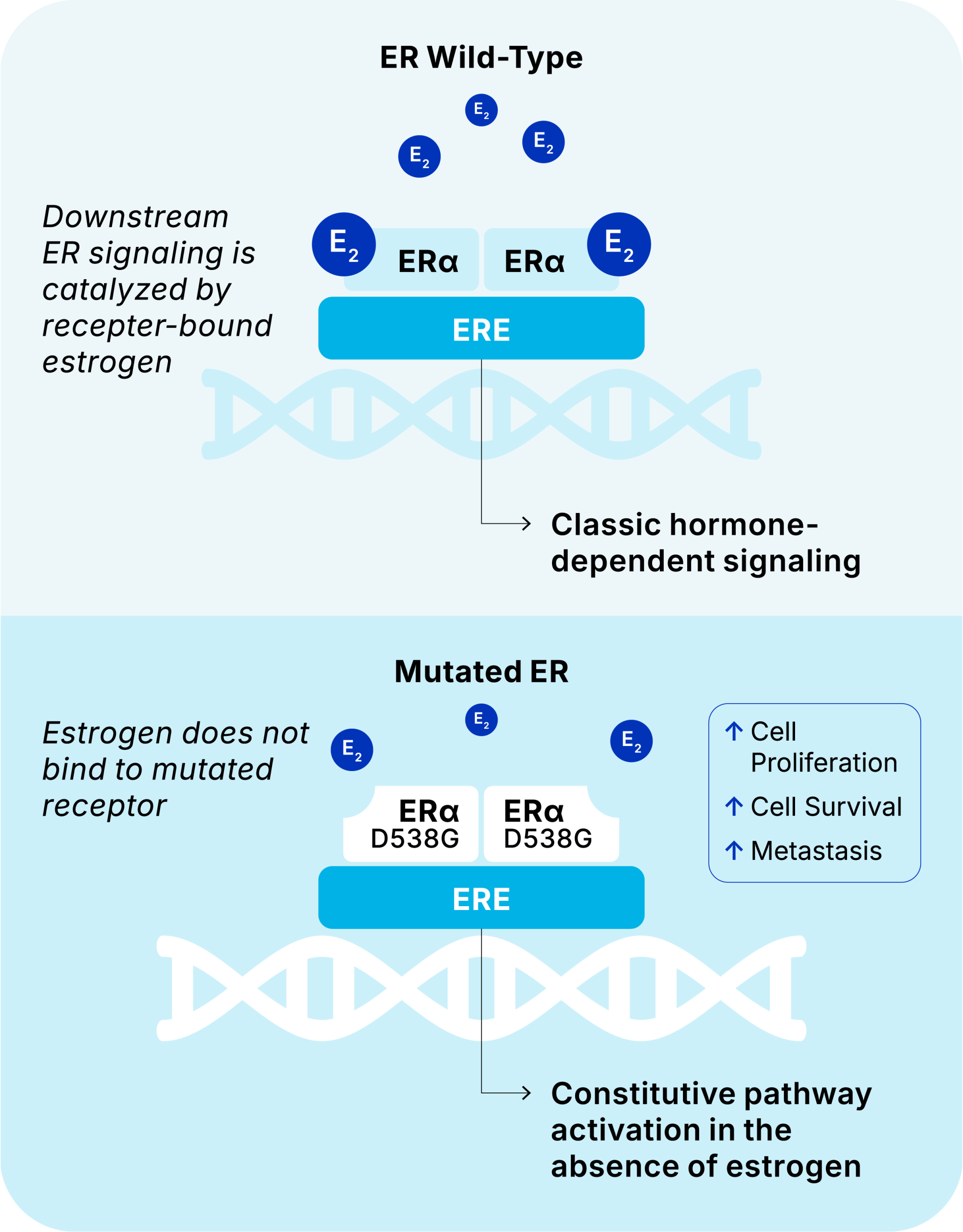
ESR1 LBD mutations confer constitutive activation of the ER signalling pathway even in the absence of bound estrogen.
This estrogen-independent disease transformation induced by ESR1 mutations therefore renders the cancer resistant to endocrine therapies which would normally limit the body’s production of estrogen, thus increasing the risk of disease progression.4
To overcome endocrine resistance caused by AI treatment, another class of drugs called selective estrogen receptor degraders (SERDs) can be prescribed to circumvent this estrogen-independent disease transformation. SERDs degrade estrogen receptors, thus blocking the activation of the ESR1 gene, making them effective even when tumors have developed resistance to AIs.1
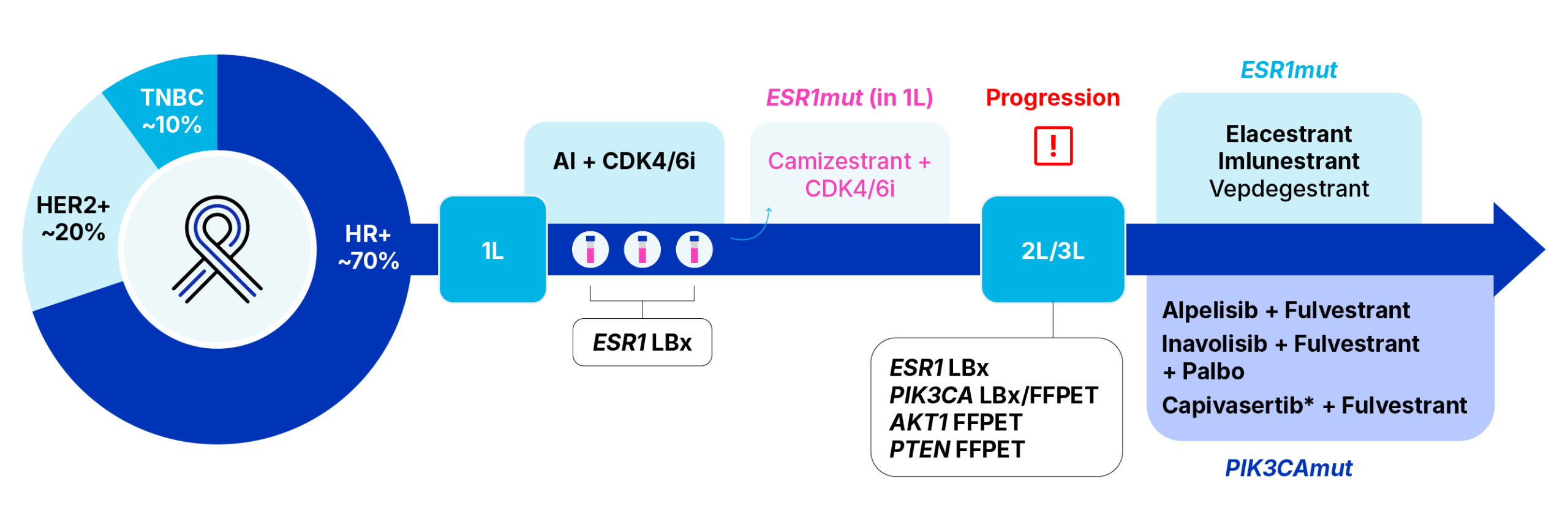
In January 2023, the US Food & Drug Administration approved elacestrant (Orserdu), the first targeted therapy for HR+ patients with mBC presenting with any one or more ESR1 mutations who have progressed following treatment with endocrine therapy.7 In the registrational, Phase III EMERALD trial, HR+ mBC patients with at least 12 months prior exposure to CDK4/6i, experienced a significant improvement in median progression-free survival (mPFS) taking monotherapy elacestrant compared to those taking fulvestrant (Faslodex), an injectable SERD, or an AI (mPFS of 8.61 vs 1.91 months, HR 0.41). More recently, in September 2025, the FDA also approved imlunestrant (Inluriyo™) as a monotherapy for patients who have experienced disease progression following treatment with an AI + CDK4/6i, highlighting an mPFS improvement of 5.5 months for imlunestrant compared to 3.8 months for patients continuing on investigator’s choice of endocrine therapy.8
Molecular Testing via Liquid Biopsy
Liquid biopsy testing presents a highly sensitive, minimally invasive means of testing for clinically meaningful ESR1 mutations without the need for surgical resection to obtain tissue biopsy.4 Since ESR1 mutations are acquired following treatment exposure and are typically subclonal in nature, testing via liquid biopsy is preferred. The archival tumour specimen is unlikely to be positive for such mutations.9,10 To date, most testing for ESR1 mutations is performed on circulating tumor DNA (ctDNA), fragments of DNA shed from tumor cells into the bloodstream via apoptosis and necrosis, in human plasma.4
Various molecular technologies, including real-time, quantitative polymerase chain reaction (qPCR), digital PCR (dPCR), and next-generation sequencing (NGS), have demonstrated the ability to accurately detect ESR1 mutations from human plasma, reaching sensitivity of ≤1% variant allele frequency (VAF).4
Emerging Need for ESR1 Mutation Surveillance
The emergence of novel, oral SERDs has cemented ESR1 mutation testing in key breast cancer clinical practice guidelines, including those curated by the National Comprehensive Cancer Network (NCCN), the American Society of Clinical Oncology (ASCO), and the European Society for Medical Oncology (ESMO).11
Learn More About the Importance of ESR1 Mutation Monitoring
ESR1 Testing and Monitoring in Metastatic Breast Cancer: A Paradigm Shift for Molecular Pathology Labs
While these guidelines currently recommend ESR1 mutation testing at clinical progression following first-line AI + CDK4/6i treatment, emerging clinical trial data is paving the way for ESR1 mutation surveillance during frontline therapy to intervene with effective therapies before a patient experiences anatomical progression, thus driving treatment decisions earlier in order to improve overall cancer care.
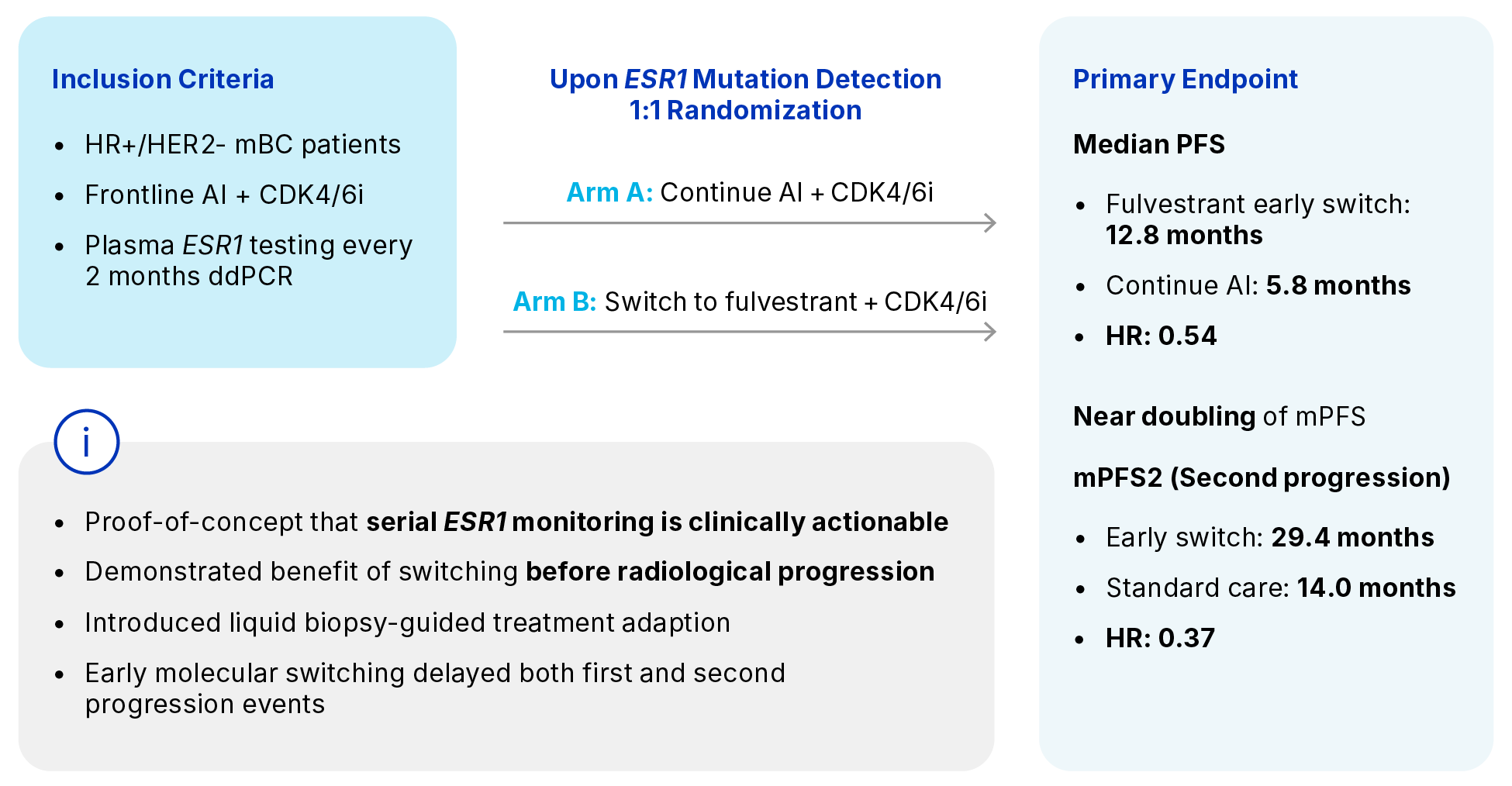
The Importance of PADA-1 Trial12
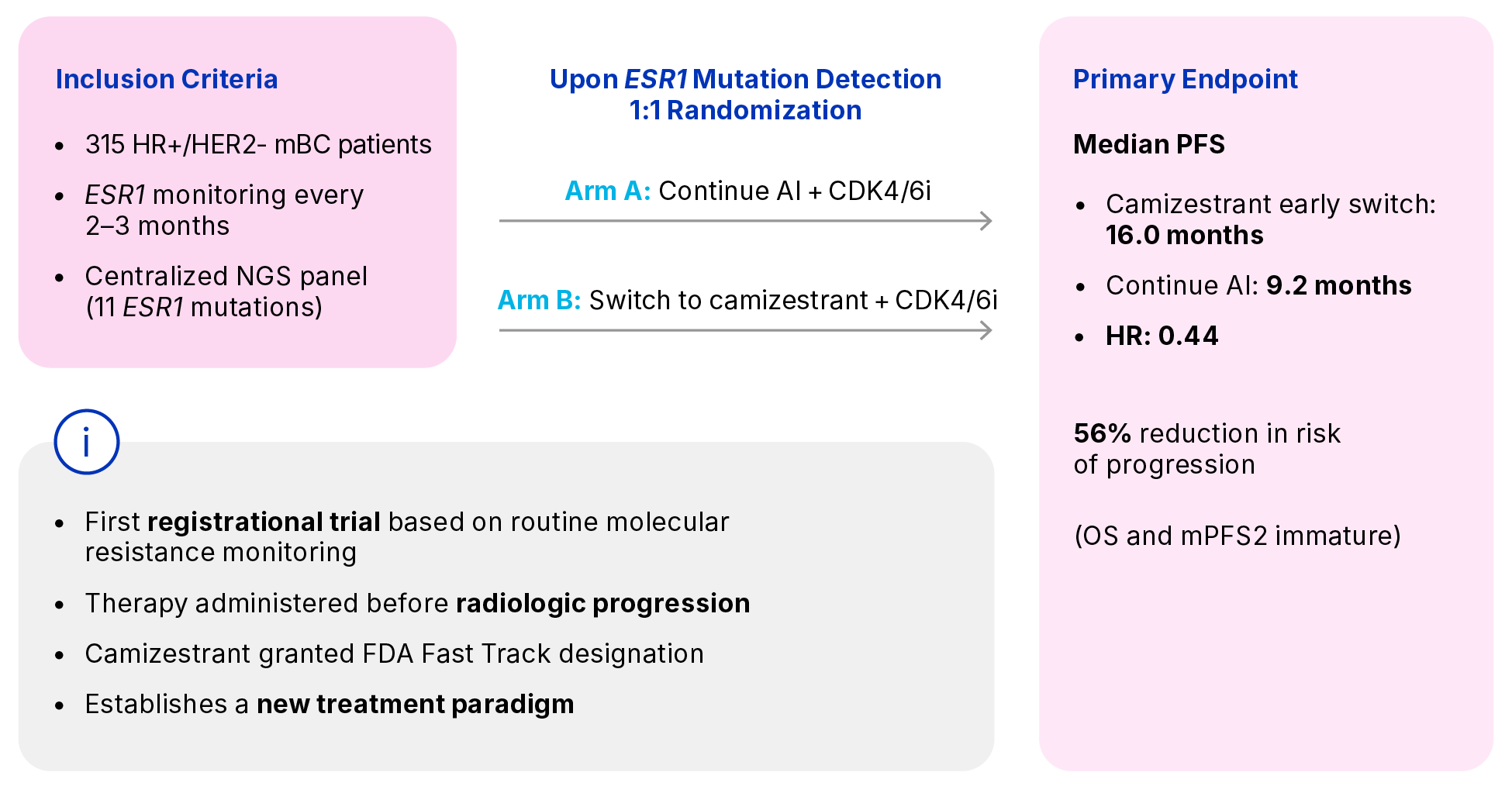
SERENA-6: A Landmark Trial in Breast Cancer Research13,14
A Shift in the Testing Paradigm in HR+/HER2- mBC
From reactive testing to proactive monitoring for biomarker-guided treatment
Old Model
Treat → Wait for progression → Switch
New Model
Treat → Monitor ESR1 in plasma → Detect resistance early → Switch before progression
References
- Lumachi F, et al. Curr Med Chem. 2013;20(5):596-604.
- Progress Report Cancer. Available from: https://progressreport.cancer.gov/diagnosis/stage Last Accessed on 10 February 2026
- Cleveland and Clinic. Available from: https://my.clevelandclinic.org/health/treatments/24685-aromatase-inhibitors Last Accessed 10 February 2026
- McCann KE, et al. NPJ Breast Cancer. 2024 Jan 19;10(1):8.
- Cancer Center Available from : https://www.cancercenter.com/cancer-types/breast-cancer/types/rare-breast-cancer-types/metastatic-breast-cancer Last Accessed 10 February 2026
- Dustin D, et al. Cancer. 2019 Nov 1; 125(21): 3714–3728.
- OncLive Available from: https://www.onclive.com/view/fda-approves-elacestrant-for-er-her2-esr1-mutated-advanced-or-metastatic-breast-cancer Last Accessed 10 February 2026
- gov Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-imlunestrant-er-positive-her2-negative-esr1-mutated-advanced-or-metastatic-breast Last Accessed 10 February 2026
- Turner NC et al. Circulating tumour DNA analysis to direct therapy in advanced breast cancer (plasmaMATCH): a multicentre, multicohort, phase 2a, platform trial. Lancet Oncol. 2020;21(10):1296-308.
- Pascual J et al. ESMO recommendations on the use of circulating tumour DNA assays for patients with cancer: a report from the ESMO Precision Medicine Working Group. Ann Oncol. 2022;33(8):750-68.
- NCCN, ESMO & ASCO guidelines NCCN Guidelines Version 4 2025: Breast Cancer https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf ASCO Guidelines: Breast Cancer https://ascopubs.org/topics/asco-guidelines/breast-cancer ESMO Guidelines: Breast Cancer https://www.esmo.org/guidelines/esmo-clinical-practice-guidelines-breast-cancer.
- CCO Independent Conference Highlights of the 2023 ASCO Annual Meeting; Lancet Oncol. 2022;23:1367.
- NC Turner, et al. N Engl J Med. 2025 Aug 7;393(6):569-580. doi: 10.1056/NEJMoa2502929.
- J Rinaldi, et al. PLoS One. 2020 May 6;15(5):e0231999. doi: 10.1371/journal.pone.0231999.
Understanding ESR1 Mutations in Advanced Breast Cancer

Although ER blockage confers durable responses, treatment resistance and failure does occur in around 40% of patients with metastatic breast cancer (mBC), resulting in disease recurrence and progression.5 The most common form of treatment resistance stems from the acquisition of point mutations in the ligand-binding domain (LBD) of the ESR1 gene,6 resulting in constitutive activation of the ER even in the absence of ER-bound estrogen.4
The Emergence of the Mutation

ESR1 LBD mutations confer constitutive activation of the ER signalling pathway even in the absence of bound estrogen.
This estrogen-independent disease transformation induced by ESR1 mutations therefore renders the cancer resistant to endocrine therapies which would normally limit the body’s production of estrogen, thus increasing the risk of disease progression.4
To overcome endocrine resistance caused by AI treatment, another class of drugs called selective estrogen receptor degraders (SERDs) can be prescribed to circumvent this estrogen-independent disease transformation. SERDs degrade estrogen receptors, thus blocking the activation of the ESR1 gene, making them effective even when tumors have developed resistance to AIs.1
In January 2023, the US Food & Drug Administration approved elacestrant (Orserdu), the first targeted therapy for HR+ patients with mBC presenting with any one or more ESR1 mutations who have progressed following treatment with endocrine therapy.7 In the registrational, Phase III EMERALD trial, HR+ mBC patients with at least 12 months prior exposure to CDK4/6i, experienced a significant improvement in median progression-free survival (mPFS) taking monotherapy elacestrant compared to those taking fulvestrant (Faslodex), an injectable SERD, or an AI (mPFS of 8.61 vs 1.91 months, HR 0.41). More recently, in September 2025, the FDA also approved imlunestrant (Inluriyo™) as a monotherapy for patients who have experienced disease progression following treatment with an AI + CDK4/6i, highlighting an mPFS improvement of 5.5 months for imlunestrant compared to 3.8 months for patients continuing on investigator’s choice of endocrine therapy.8
Molecular Testing via Liquid Biopsy
Liquid biopsy testing presents a highly sensitive, minimally invasive means of testing for clinically meaningful ESR1 mutations without the need for surgical resection to obtain tissue biopsy.4 Since ESR1 mutations are acquired following treatment exposure and are typically subclonal in nature, testing via liquid biopsy is preferred. The archival tumour specimen is unlikely to be positive for such mutations.9,10 To date, most testing for ESR1 mutations is performed on circulating tumor DNA (ctDNA), fragments of DNA shed from tumor cells into the bloodstream via apoptosis and necrosis, in human plasma.4
Various molecular technologies, including real-time, quantitative polymerase chain reaction (qPCR), digital PCR (dPCR), and next-generation sequencing (NGS), have demonstrated the ability to accurately detect ESR1 mutations from human plasma, reaching sensitivity of ≤1% variant allele frequency (VAF).4
Emerging Need for ESR1 Mutation Surveillance
The emergence of novel, oral SERDs has cemented ESR1 mutation testing in key breast cancer clinical practice guidelines, including those curated by the National Comprehensive Cancer Network (NCCN), the American Society of Clinical Oncology (ASCO), and the European Society for Medical Oncology (ESMO).11
Learn More About the Importance of ESR1 Mutation Monitoring
ESR1 Testing and Monitoring in Metastatic Breast Cancer: A Paradigm Shift for Molecular Pathology Labs
While these guidelines currently recommend ESR1 mutation testing at clinical progression following first-line AI + CDK4/6i treatment, emerging clinical trial data is paving the way for ESR1 mutation surveillance during frontline therapy to intervene with effective therapies before a patient experiences anatomical progression, thus driving treatment decisions earlier in order to improve overall cancer care.
The Importance of PADA-1 Trial12
SERENA-6: A Landmark Trial in Breast Cancer Research13,14
A Shift in the Testing Paradigm in HR+/HER2- mBC
From reactive testing to proactive monitoring for biomarker-guided treatment
Old Model
Treat → Wait for progression → Switch
New Model
Treat → Monitor ESR1 in plasma → Detect resistance early → Switch before progression
References
- Lumachi F, et al. Curr Med Chem. 2013;20(5):596-604.
- Progress Report Cancer. Available from: https://progressreport.cancer.gov/diagnosis/stage Last Accessed on 10 February 2026
- Cleveland and Clinic. Available from: https://my.clevelandclinic.org/health/treatments/24685-aromatase-inhibitors Last Accessed 10 February 2026
- McCann KE, et al. NPJ Breast Cancer. 2024 Jan 19;10(1):8.
- Cancer Center Available from : https://www.cancercenter.com/cancer-types/breast-cancer/types/rare-breast-cancer-types/metastatic-breast-cancer Last Accessed 10 February 2026
- Dustin D, et al. Cancer. 2019 Nov 1; 125(21): 3714–3728.
- OncLive Available from: https://www.onclive.com/view/fda-approves-elacestrant-for-er-her2-esr1-mutated-advanced-or-metastatic-breast-cancer Last Accessed 10 February 2026
- gov Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-imlunestrant-er-positive-her2-negative-esr1-mutated-advanced-or-metastatic-breast Last Accessed 10 February 2026
- Turner NC et al. Circulating tumour DNA analysis to direct therapy in advanced breast cancer (plasmaMATCH): a multicentre, multicohort, phase 2a, platform trial. Lancet Oncol. 2020;21(10):1296-308.
- Pascual J et al. ESMO recommendations on the use of circulating tumour DNA assays for patients with cancer: a report from the ESMO Precision Medicine Working Group. Ann Oncol. 2022;33(8):750-68.
- NCCN, ESMO & ASCO guidelines NCCN Guidelines Version 4 2025: Breast Cancer https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf ASCO Guidelines: Breast Cancer https://ascopubs.org/topics/asco-guidelines/breast-cancer ESMO Guidelines: Breast Cancer https://www.esmo.org/guidelines/esmo-clinical-practice-guidelines-breast-cancer.
- CCO Independent Conference Highlights of the 2023 ASCO Annual Meeting; Lancet Oncol. 2022;23:1367.
- NC Turner, et al. N Engl J Med. 2025 Aug 7;393(6):569-580. doi: 10.1056/NEJMoa2502929.
- J Rinaldi, et al. PLoS One. 2020 May 6;15(5):e0231999. doi: 10.1371/journal.pone.0231999.