Healthcare Provider Resources: Genetic Testing for Fragile X
Fragile X Syndrome Info for Healthcare Providers
Fragile X syndrome
Fragile X syndrome (FXS) is the most common known inherited cause of intellectual disability and autism spectrum disorder. About half of the males with FXS also have autism, and one out of every five females with FXS have autism. FXS testing may be relevant to as many as 400,000 children in the US.
Genetics of Fragile X Syndrome
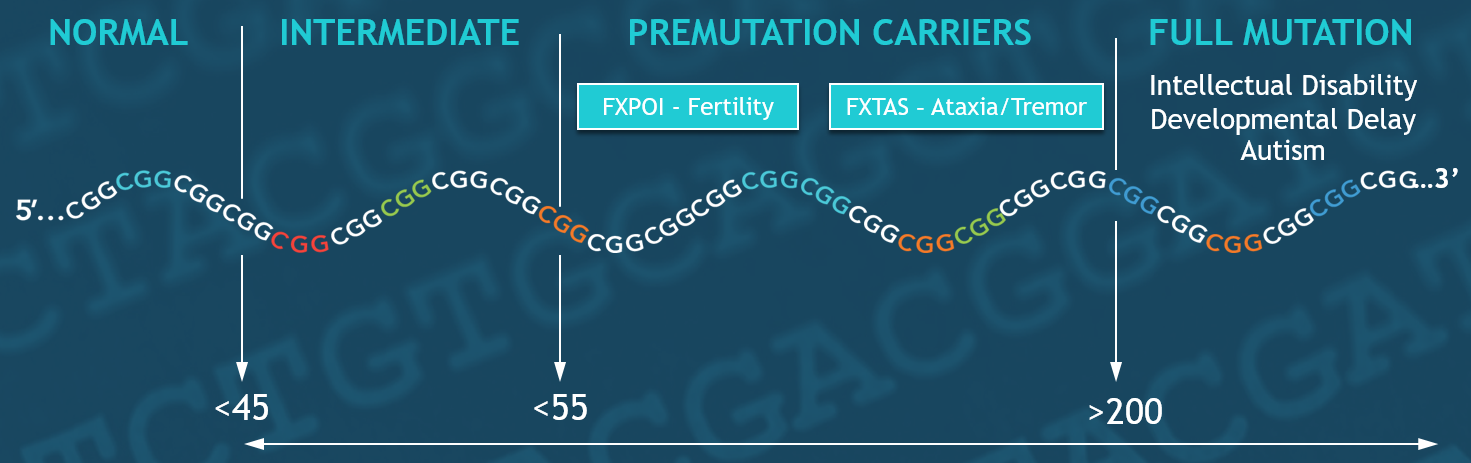
FXS is caused by a full mutation in the FMR1 gene. FMR1 alleles can be categorized based on the number of cytosine-guanine-guanine (CGG) repeats in the 5′ promoter region of the FMR1 gene (Table 1). The clinical categories that are associated with clinical features are premutations and full mutations. Full mutations have more than 200 CGG repeats; premutations have 55 to 199 CGG repeats.
Table 1: Clinical categories and carrier status of mutations in the FMR1 gene.
| FMR1-CGG REPEAT NUMBER | Clinical CATEGORY | CLINICAL FEATURES | Carrier Status and Stability of FMR1 CGG-Repeat region |
| 5-44 | Normal | None | Not a carrier of FXS.
Stable allele. Allele passed from parent to child with rare changes in repeat size. |
| 45-54 | Intermediate | None | Not a carrier.
Stable allele. Alleles occasionally undergo a small change in repeat size in some families. |
| 55-199 | Premutation | FXTAS symptoms over age 50
FXPOI in women |
Carrier of FXS
Unstable allele with risk of large changes in repeat size from one generation to the next which can result in a full mutation and FXS. Risk of expansion varies based on total repeat number of CGG repeats and AGG interruptions. |
| ≥200 | Full mutation | Fragile X syndrome with or without autism | Unstable allele |
Clinical features of FXS
Clinical features of FXS vary widely but can include mild to severe cognitive delays, learning disability, or math disability, and intellectual disability of unknown etiology, and autism spectrum disorder including Pervasive Developmental Disorder-Not Otherwise Specified (PDD-NOS) or Asperger syndrome, and seizures.
Behavioral features of FXS also include a broad range of issues, such as perseverative or repetitive speech, hand flapping, poor eye contact, anxiety, attention problems, and hyperactivity.
Physical features that are often associated with FXS are a long narrow face, large ears, large head, hyperflexible joints, prominent jaw and forehead, and large testes in males after puberty. The heart may have a mitral valve prolapse, which is usually not life-threatening, but in severe cases, surgery might be required to correct the problem.
Figure 1. Impact of AGG interruptions on risk of expansion to Full Mutation
FMR1 premutation-associated disorders
Testing for an FMR1 premutation is warranted if the patient has symptoms associated with fragile X-associated tremor ataxia syndrome (FXTAS) or fragile X-associated primary ovarian insufficiency (FXPOI), and/or family history of FXS or FXTAS or FXPOI. Some people with premutations are also at risk for learning problems, autism spectrum features, attention deficits, and depression or anxiety disorders.
FX-associated primary ovarian insufficiency (FXPOI)
Women who have a premutation are at risk for FXPOI. Common symptoms of FXPOI include irregular menstrual cycles and early menopause by the time they are 40 years old. Some women may become infertile, but some women with FXPOI can still get pregnant. FXPOI is common, one out of four or five women who are carriers experience FXPOI.
FX-associated tremor ataxia syndrome (FXTAS)
Men and women who have a premutation are at risk for FX-associated tremor ataxia syndrome (FXTAS), although FXTAS occurs more often in men than women. Symptoms of FXTAS appear as a person ages, usually over 50 years old (see Clinical Features of FXTAS, inset box).
Diagnosis of FXS and FMR1-associated disorders
Molecular testing of the fragile X gene (FMR1) for mutations is the standard, first-tier approach for FXS and for unexplained developmental delay, intellectual disability, and autism. Testing identifies mutations by determining the number of CGG repeats in the FMR1 gene, and reports the repeat number(s) and classifies the gene into clinical categories: full mutation, premutation, intermediate, and normal (Table 1). FMR1 is X-linked, therefore, an FX test result will generate one CGG-repeat number for males, and two numbers for females, one for each allele. Testing can also uncover mosaicism, which can detect additional repeat alleles otherwise missed with conventional approaches. Additional methylation analysis of the FMR1 gene can be performed to understand the allele-specific level of activity of the gene, which can impact a prognosis.** People with size and/or methylation mosaicisms may have milder symptoms than individuals with completely methylated full mutations.***
FMR1 testing offers the highest diagnostic yield of any single gene in autism molecular diagnostics. For FXS testing, it is important to have an assay that can accurately to determine the CGG repeat numbers.
In February of 2020, the United States Food and Drug Administration (FDA) cleared the first laboratory test (AmplideX® Fragile X Dx and Carrier Screen Kit) to aid in the diagnosis of fragile X syndrome and FMR1-associated disorders [including FX-associated primary ovarian insufficiency (FXPOI) and FX-associated tremor ataxia syndrome (FXTAS)]. Asuragen developed a test that reliably identifies all clinical categories and accurately reports repeat numbers for FMR1 expansions. The test is provided to CAP/CLIA-accredited laboratories that perform genetic testing. Ask your laboratory if they are using the AmplideX® test for fragile X testing.
In addition, FMR1 testing detects low level mosaics across all clinical categories with more sensitively than other testing approaches such as Southern blot. The AmplideX® test is also indicated for use in screening asymptomatic carriers at-risk for having children with fragile X syndrome. Click here to learn more.
Carrier screening & risk assessment
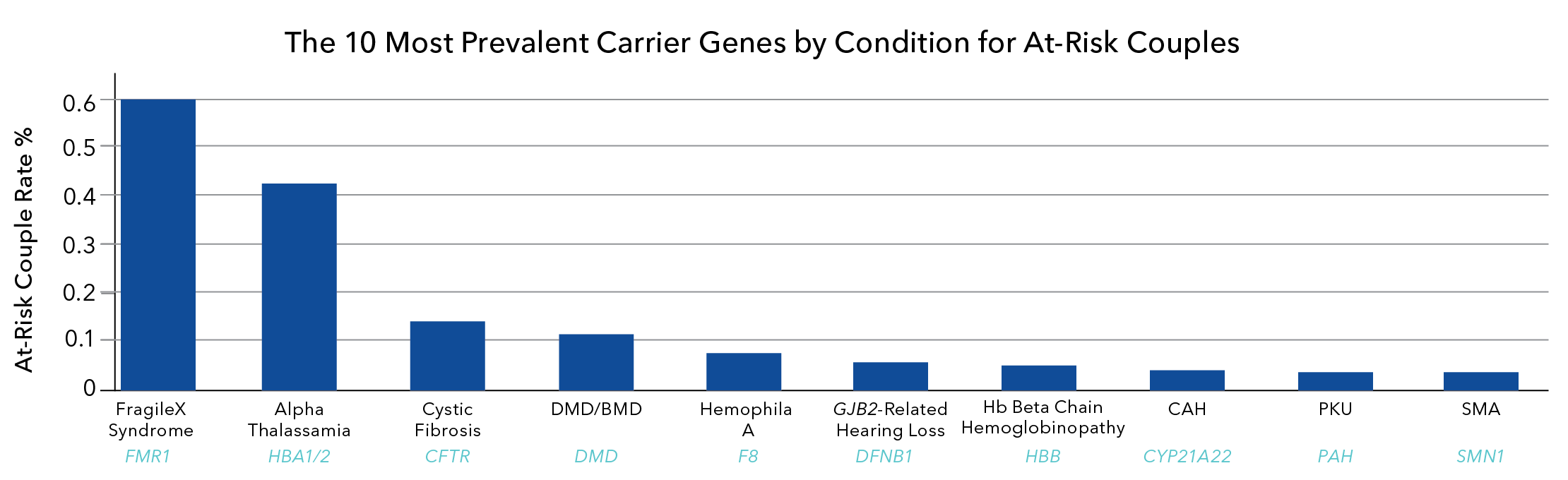
Fragile X syndrome FXS is transmitted as an X-linked disorder, and the condition is caused by a full mutation expansion (>200 CGG repeats) in the FMR1 gene. Carrier screening for FXS is recommended by the American College of Obstetricians and Gynecologists (ACOG). FXS has one of the highest genetic carrier rates across populations, and more than one million women are estimated to be fragile X carriers. Carriers may be asymptomatic.
Figure 2. At-risk couple rate percent is highest for fragile X syndrome****, indicating that fragile X carrier screening would be the most helpful in identifying at-risk couples.
Different clinical categories for fragile X are listed in Table 1. Normal alleles (~5 to 44 FMR1-CGG repeats) are stable when passed from parent to child. Intermediate alleles (45–54 FMR1-CGG repeats) occasionally undergo small changes in repeat size in some families during transmission. Premutation alleles (55–199 FMR1-CGG repeats) are unstable with large changes in repeat size from one generation to the next, which may result in full mutation expansions and the FXS.
Fragile X carriers have an FMR1-premutation (Table 1). For carriers, there is a risk for the premutation in the mother to expand to a full mutation in their child. The larger the size of the premutation, the higher the risk for passing on an expanded full mutation. An additional genetic test can refine the risk of having a child with FXS by determining the number of AGG repeats in the FMR1-CGG repeat tract.
Risk assessment
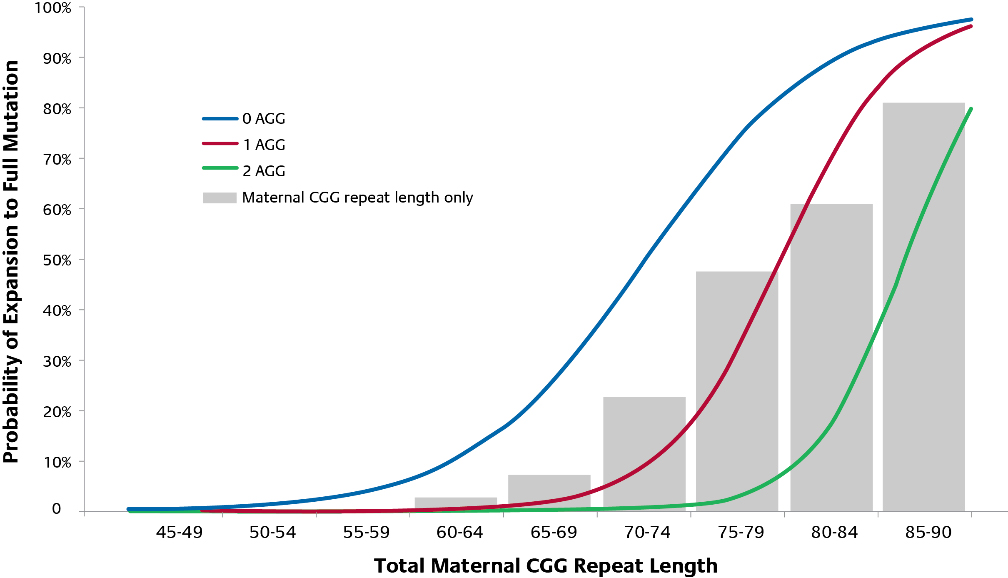
Genetic FXS testing enables routine carrier screening to identify premutations, and to report CGG repeat numbers for premutations. The number of CGG repeats in a premutation can be used to assess the risk of repeat expansion from parent to child. Additional testing to identify AGG interruptions can further personalize the risk of full-mutation expansion from parent to child. The likelihood of expansion to a fragile X full mutation is based on the total number of CGG repeats and the number of AGG interruptions. As illustrated in the graph (Figure 3), the grey bars represent the risk of expansion to a full mutation in the absence of knowledge of number of AGG interruptions. The lines represent the modified risk when AGG information is incorporated. As an example, an FMR1 allele with 75-79 CGG repeats has a 10-fold greater risk of expansion with 0 AGG interruptions (74%) compared to 2 AGG interruptions (7%). *****
Figure 3. Impact of AGG interruptions on risk of expansion to Full Mutation
What are the testing options?
AmplideX® Fragile X Dx and Carrier Screen Kit is the only FDA-cleared test indicated as an aid in screening asymptomatic carriers at-risk for having children with fragile X syndrome. The diagnostic kit reliably determines the number of CGG repeats in the FMR1 gene to aid in the diagnosis of FXS and associated disorders including FXTAS and FXPOI. The test also has software that has been clinically validated to provide results based on advanced machine-learning technology which reduces the risk of errors with manual interpretation. More information about the first and only fragile X test cleared by the FDA can be found at this link: AmplideX® Fragile X Dx & Carrier Screen Kit (developed by Asuragen, Inc.).
Clinical research testing for AGG interruptions in the FMR1-repeat tract has been shown to provide additional information to refine risk estimates for premutation carriers. Clinical research testing options are available and are supported by recent scientific evidence (see Publications), however the recommendations have not yet been incorporated into the guidelines. Follow this link for more information on Asuragen’s Xpansion Interpreter® service for the accurate detection of AGG interruptions.
More information about AGG interruptions
Publications
Absence of AGG Interruptions Is a Risk Factor for Full Mutation Expansion Among Israeli FMR1 Premutation Carriers. Domniz N, et al. Front Genet. 2018 Dec 13;9:606. doi: 10.3389/fgene.2018.00606. eCollection 2018.PMID: 30619448.
AGG interruptions within the CGG repeat area are protective and decrease the risk of intergenerational expansion. The authors recommended that risk estimates for FMR1 premutation carriers be based on AGG interruptions and repeat size.
Expansions and contractions of the FMR1 CGG repeat in 5,508 transmissions of normal, intermediate, and premutation alleles. Nolin SL, et al. Am J Med Genet A. 2019 Jul;179(7):1148-1156. doi: 10.1002/ajmg.a.61165. Epub 2019 May 2.PMID: 31050164.
Instability of the FMR1 repeat is observed in transmissions of premutation alleles (55-200 repeats), is influenced by the size of the repeat, its internal structure and the sex of the transmitting parent. The authors propose a refined model of FMR1 repeat progression from normal to premutation size and suggest that most normal alleles without AGG interruptions are derived from contractions of maternal premutation alleles.
The role of AGG interruptions in fragile X repeat expansions: a twenty-year perspective. Latham GJ, et al. Front Genet. 2014 Jul 29;5:244. doi: 10.3389/fgene.2014.00244. eCollection 2014.PMID: 25120560.
This publication reviews relevant studies published since 1993 on AGG interruptions and examine those publications that have shown that AGG interruptions refine risk estimates for repeat instability and full-mutation expansions.
Personalizing Risk for FMR1 Premutation Carriers: Insights from over 8000 AGG Test Results. Eveleigh, D. et al. National Fragile X Foundation Meeting, April 2019, DOI:10.13140/RG.2.2.25479.04005
Fragile X Syndrome Resources for Healthcare Providers
Footnotes
-
*
-
**
High-resolution Methylation Polymerase Chain Reaction for Fragile X Analysis: Evidence for Novel FMR1 Methylation Patterns Undetected in Southern Blot Analyses Chen, L. et al. Genet Med. 2011 Jun;13(6):528-538. doi: 10.1097/GIM.0b013e31820a780f
-
***
ACMG Standards and Guidelines for Fragile X Testing: A Revision to the Disease-Specific Supplements to the Standards and Guidelines for Clinical Genetics Laboratories of the American College of Medical Genetics and Genomics Genet Med
Monaghan, K. et al. 2013 Jul;15(7):575-86. doi: 10.1038/gim.2013.61.
https://www.acmg.net/docs/ACMG_SG_For_Fragile_X_Testing_7.24.13.PDF
-
****
Based on combined data from Ben-Shachar R, et al. Genet Med. 2019 Sep;21(9):1931-1939. and Capalbo A, et al. PLoS Genet. 2019 Oct 7;15(10):e1008409
-
*****
- Fragile X AGG analysis provides new risk predictions for 45-69 repeat alleles. Nolin, S. et al. Am J Med Genet 2013; 161(4):771-8. doi: 10.1002/ajmg.a.35833
https://pubmed.ncbi.nlm.nih.gov/23444167/ - Fragile X full mutation expansions are inhibited by one or more AGG interruptions in premutation carriers. Nolin, S. et al. Genet Med 2015 May;17(5):358-64.
https://pubmed.ncbi.nlm.nih.gov/25210937/
- Fragile X AGG analysis provides new risk predictions for 45-69 repeat alleles. Nolin, S. et al. Am J Med Genet 2013; 161(4):771-8. doi: 10.1002/ajmg.a.35833